
NAFDAC warns against ‘counterfeit’ cancer drug in circulation
The National Agency for Food and Drug Administration and Control (NAFDAC) has alerted Nigerians, including healthcare providers, about Phesgo, a suspected counterfeit cancer treatment drug.
The alert on the circulation of the drug was published on the agency’s website.
NAFDAC said a doctor at the Lagos University Teaching Hospital (LUTH) had raised concerns about the drug after a patient brought it in.
The agency said the product had not been administered, but it matched the characteristics of a previously reported counterfeit batch, C3809C51.
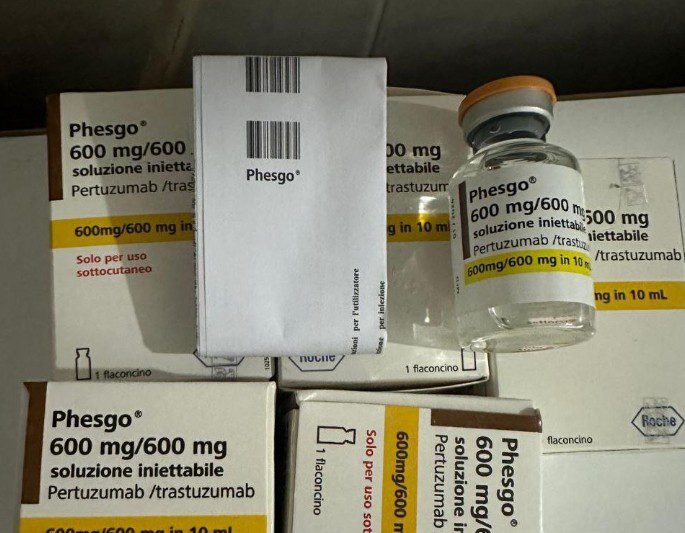
“The National Agency for Food and Drugs Administration and Control (NAFDAC) wishes to inform healthcare providers and the public of a report of a suspected counterfeit Phesgo® 600mg/600mg/10ml, labelled with batch C5290S20,” the statement reads.
“The Marketing Authorisation Holder (MAH) Roche received a complaint from a doctor at Lagos University Teaching Hospital (LUTH-NSIA) reporting a suspected counterfeit Phesgo® 600mg/600mg, labelled with batch C5290S20.
“The product was reported to have been brought in by a patient for administration. It had not been administered at the time of the report, as it matched the previously reported counterfeit batch: C3809C51.
“Although no sample was returned to Roche for investigation, only pictures displaying parts of a Phesgo® 600mg/600mg in a 10ml folding box and a labelled vial.
“Images of the suspected product were examined by Roche and compared to the genuine samples retained for reference.
“Although no physical sample was returned to Roche for investigation, images of parts of the product specifically, a Phesgo 600mg/600mg vial and a 10ml folding box were examined.
“The suspected product’s images were compared to genuine samples retained by Roche.
“Roche’s investigation identified several significant differences between the complaint sample and genuine materials, confirming the counterfeit status of the batch.
“These included: no batch number in the MAH database, discrepancies in language, missing basilisk, incorrect bollino date, and tampered evidence labels that did not match genuine Roche materials.”
NAFDAC added that “since no physical sample was available for chemical analysis”, the investigation was “limited to visual comparisons”.